Photochemistry and Photobiology
Because of the strong ozone absorption, any photons with wavelengths shorter than 280 nm at ground level are most likely due to human activity (or lightning). This is just as well since these short wavelength photons have enough energy to break many chemical bonds. We have found many uses for this bond breaking capability in material processing. Irradiation with 254 nm radiation is useful for cleaning organics from optical surfaces and from semiconductor wafers. "Germicidal radiation" (UVC) is used for sterilization and germicidal lamps can still be found in some European meat shops. UV curing is extensively employed in industry and dentistry. Current DOE (Department of Energy) sponsored work at the National Renewable Energy Laboratory in Golden, CO, aims at using the aggressive nature of ultraviolet (solar) radiation to detoxify hazardous wastes.
There is a concern about the observed increase in UVB because of the effect of UVB on many important biological molecules. We cannot yet assess the severity of the potential problem because of the shortage of reliable measurements of the ultraviolet loading, and because of continued uncertainty about the impact of ultraviolet.
The Uncertain UV; Definition of UVA, UVB and UVC
UV radiation offers many technical challenges. Transmittance and refractive index of many optical materials change rapidly through the ultraviolet. Detectors and optical coatings, and even some UV filter materials (see Figure 2) are not stable as the high energy photons cause changes. Even the definition of the UVA, UVB, and UVC is in dispute. The Commission Internationale d'Eclairage (CIE), the world authority on definitions relating to optical radiation, changes from tradition in defining the regions.
Table 1 Definitions of UV Regions
| UVC | UVB | UVA | |
|---|---|---|---|
| CIE | 100 - 280 nm | 280 - 315 nm | 315 - 400 nm1 |
| Traditional Versions | 200 - 290 nm | 290 - 320 nm | 320 - 400 nm2 |
| This Tutorial | <280 nm | 280 - 320 nm | 320 - 400 nm3 |
1) International Lighting Vocabulary, CIE Publ. No. 17.4
2) UVA Biological Effects of Ultraviolet Radiation with Emphasis on Human Responses to Longwave Ultraviolet, Parrish et al. Plenum Press 1978
3) Influences of Atmospheric Conditions and Air Mass on the Ratio of Ultraviolet to Solar Radiation, Riordan, C et al. SERI/TP 215 3895. August 1990
Most instrumentation to measure UVB in use in the U.S., and most publications, use one of the traditional definitions. Meyrick and Jennifer Peak of Argonne National Laboratory have argued persuasively that there are good reasons to retain the historic 320 nm boundary between UVB and UVA. The differences may seem small but are very significant. 5 nm is 14% of the total UVB range, but the rapid fall-off of terrestrial solar UV in the 290 - 320 nm wavelength range gives disproportionate significance to the location of these boundaries. It is important to know what definition any publication or meter is using. Here we use:
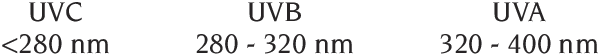
Measurement of Solar and Simulator UV
For precision quantitative work, detailed spectro-radiometric measurements are preferred over data from UVB or UVC meters. When you use a broadband meter, with a filter to exclude all but the UVB irradiance, the spectral distribution of the calibration source must be a good match to that of the unknown. Repeated independent studies by Diffey and Sayre have shown that using calibrated broadband meters can lead to huge errors because of mismatch of calibration and measured spectra.
Spectroradiometry is more complicated than using a simple meter. The very steep fall-off of terrestrial UV (see Figure 3) puts stringent demands on UV spectroradiometers used to measure the radiation below 300 nm; for accuracy the instrument requires a well characterized instrumental spectral function, exact spectral calibration and wide dynamic range without the usual problem of scattered longer wavelength radiation. A 1 nm error in calibration makes little difference in the visible but at 295 nm, 0.1 nm corresponds to a 10% difference in recorded solar irradiance. We use the accurate 253.7, 289.4, 302.2 and 337 nm lines from our spectral calibration lamps to ensure wavelength accuracy. We use filter techniques and solar blind detectors to ensure that the holographic gratings in our 77274 Double Monochromator have adequate rejection. You cannot achieve high accuracy even with the best instrumentation and care, because UV calibration standards are limited to a few percentages of absolute accuracy.
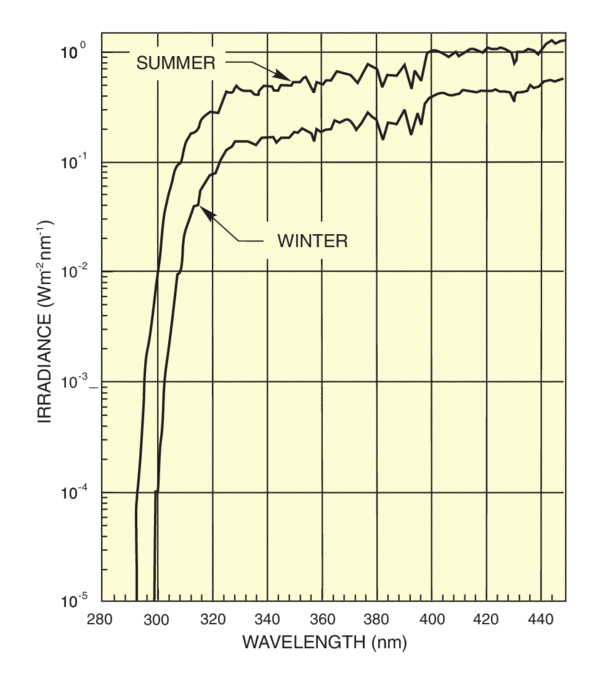
Simulation of the Solar UV
Biological testing requires accurate simulation of the solar UV especially the UVB region. One problem is that there is no accepted standard data set for solar UVB.
The data in ASTM 891 and 892 is calculated from the E 490 standard using sophisticated models for atmospheric radiation transfer. The table below shows the values for the lowest wavelengths covered by the standards; the ASTM standards are obviously not adequate for the 280 - 320 nm region.
Table 2 Irradiance Values for Lowest Wavelengths Covered by ASTM Standards
| Lowest Wavelengths (nm) | ASTM 891 AM 1.5 D Irradiance (W m-2 nm-1) | ASTM 892 AM 1.5 G Irradiance (W m-2 nm-1) | CIE AM 1 D Irradiance (W m-2 nm-1) |
|---|---|---|---|
| 305 | 0.0034 | 0.0092 | 0.0241 |
| 310 | 0.0156 | 0.0408 | 0.0683 |
| 315 | 0.0411 | 0.1039 | 0.135 |
| 320 | 0.0712 | 0.2379 | 0.1999 |
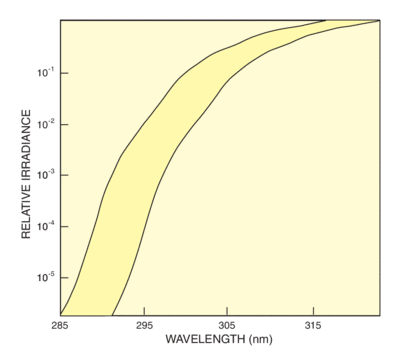
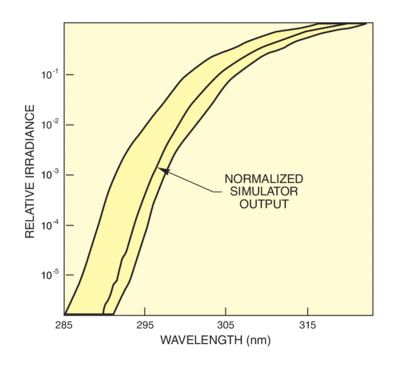
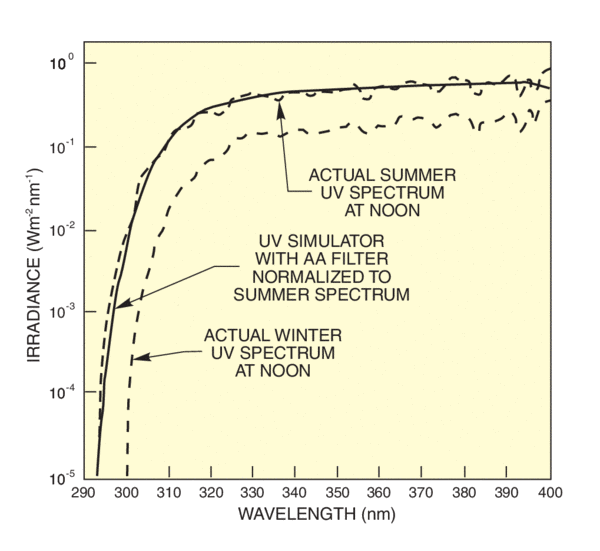
To meet the needs of researchers in the cosmetics industry for a usable standard, the CIE accepted a proposal from The Sunscreen High SPF Working Group of the Cosmetic, Toiletry and Fragrance Association. This proposal defines an acceptance band criterion for simulators used for testing sunscreen efficacy. The spectral output of the simulator below 320 nm must fall within two curves separated by 6 nm. Figure 4 shows the acceptance band, which is based in Albuquerque, NM. Figure 5 also shows how we meet this requirement with our UV Simulators and Atmospheric Attenuation filter. Simulators matching this standard allow meaningful laboratory testing of sunscreen protection factors for the UVB. Extension of the standard to the UVA is required because of the growing recognition of the dangers of longer wave UV.
Reduced Visible/Infrared Simulator
UV constitutes about 3.5% of an AM 1D simulator output. Higher UV radiation levels speed studies of UV effects. However, the higher UV levels from a conventional simulator are accompanied by proportionally more visible and infrared. Biological samples that are normally just warmed by normal solar radiation levels can be heated above viability by an intense simulator. Less drastic thermal effects may mask the true UV dependence of an effect under investigation. Our Solar UV Simulators remove most of the visible and infrared (85%), allowing exposure with UV levels many times above solar levels.
UV Filters
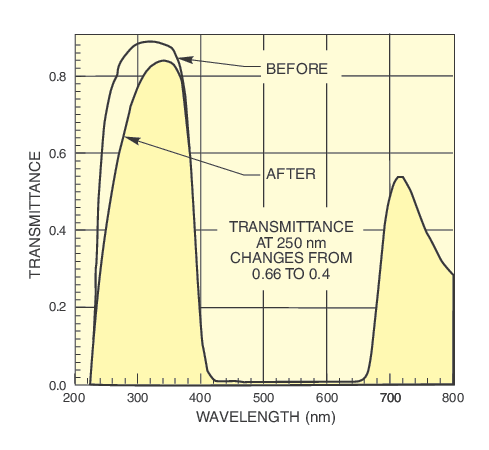
Long pass filters remove shortwave UV. Transmittance falls rapidly to negligible values below the cut-on. Figure 7 shows the transmittance of the filters we use to mimic the atmospheric transmittance, to cut out UVC and UVC plus UVB. Our comprehensive range of broadband and narrowband filters allows versatility in selection of output spectrum. The collimated beam from our simulators simplifies filter design.
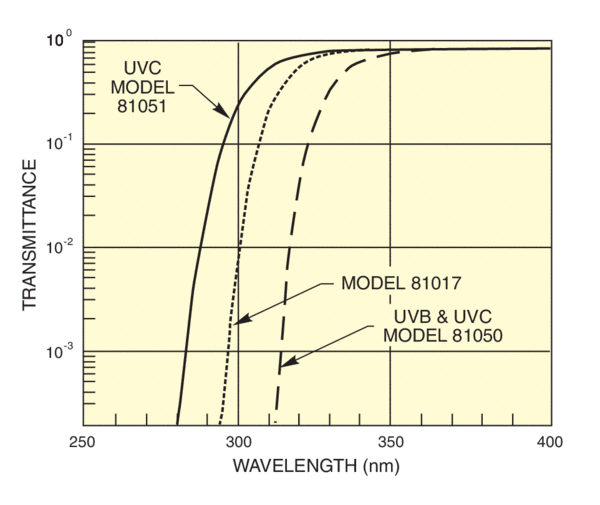
Note: any filter for use in a simulator with UV output should be stabilized by UV exposure. The order of filter positioning should be considered; highly absorbing filters should be farthest from the source. If UV is not required, then a suitable long pass filter will protect subsequent optics and simplify safety requirements.
Some Biological Effects of Light
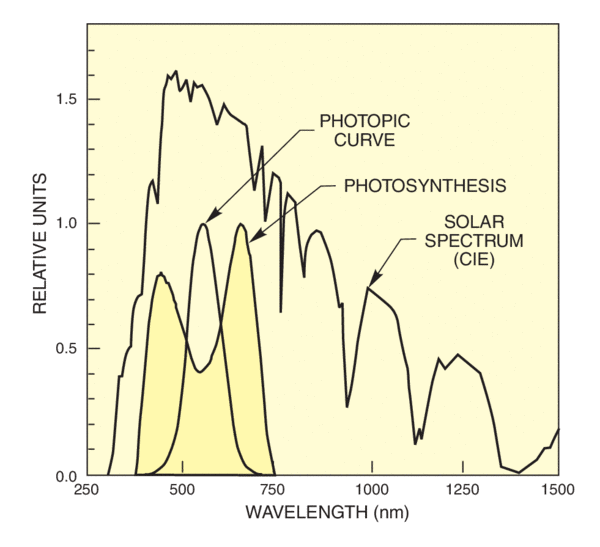
Radiation has benign and harmful effects on biological systems. Photosynthesis is obviously of vital importance; other benign effects include the production of vitamin D3, the setting of mood and the circadian rhythms, and the benefits from the mild germicidal bath provided by the sun.
There are many harmful effects of solar radiation on humans, particularly of UV radiation. Skin cancer, cataracts, and loss of skin elasticity, erythema, suppression of the immune system, photokeratitis and conjunctivitis can all result from UV exposure, though in many cases the precise relationship between exposure and effect remains unclear. Simultaneous irradiance with different wavelengths enhances some processes. Understanding how solar radiation effects plant and plankton growth is also important in assessing the results of environmental change.
Key Action Spectra
The action spectrum characterizes the wavelength dependence of a specified biological change. Researchers continue to measure action spectra for important biological processes, leading to better understanding of the effects of irradiation and potential changes due to ozone layer depletion. Knowledge of action spectra helps in the development of protective agents. Action spectra for various detrimental ultraviolet effects were used to compile the maximum recommended exposure graph.
Current research efforts include studies of the relationship between monochromatic and broadband action spectra, and better understanding of photoaddition, photorecovery and the questionable photoaugmentation.
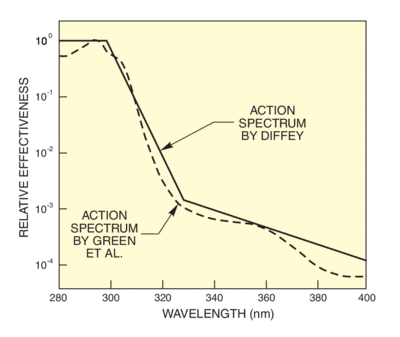
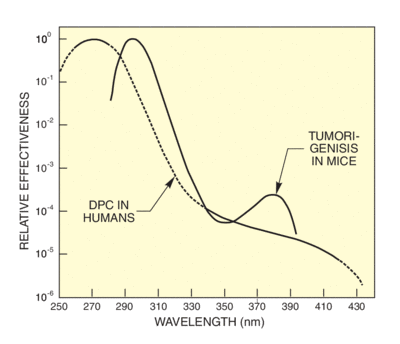
Here we show action spectra for erythema, for carcinogenisis and for DNA changes and photosynthesis inhibition in plant life. All of these spectra peak below 300 nm, but have measurable values through the UVA.
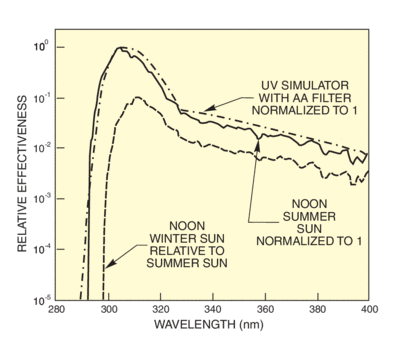
There are several established action spectra for erythema. This is understandable since there is no "standard skin" and measurements indicate that spectra differ depending on the delay from exposure to assessment. Diffey's spectrum has an uncomplicated mathematical formula that simplifies determination of the effective erythemal dose, given the solar or simulator spectrum. We use Diffey's formula to calculate effectiveness spectra for solar UV and our UV simulator. The effectiveness spectra in Figure 8 use Diffey's formula and the sun and simulator spectra from Figure 6. Figure 9 shows wavelengths actually produce erythema, taking into account the action spectrum and the availability of radiation at each wavelength. You can see that the peak effectiveness is at 305 nm for the summer sun. This is the wavelength at which the rapidly rising solar spectrum compensates the falling action spectrum for maximum effect. In the winter, the effectiveness is much reduced and the peak shifted to longer wavelengths.
The UTR5 spectrum for tumorigenisis in hairless mice and the action spectrum for DNA damage in human cells indicate a strong deep UV dependence. Like all the action spectra we show, these have been normalized and do not indicate absolute sensitivity to radiation level. Quantification of actual sensitivity in the case of tumor induction is complicated not only by the statistical nature of cancer development, but also by shielding factors. In vitro irradiated cells do not have the in vivo shielding of epidermal layers.
Figure 11 shows two action spectra for photosynthesis inhibition in Antarctic phytoplankton (drawn from Helbling4 and Mitchell5). Unlike tropical phytoplankton, Antarctic phytoplankton, particularly dark-adapted (subpynocline) phytoplankton from greater depths, are strongly affected by UV radiation. The sensitivity and photoadaptability of this basic material will influence the impact of the Antarctic ozone hole on the local food web. Long pass filtering of solar radiation was used to determine both of these spectra; differences may be due to sampling technique (e.g. sample depth) or the low spectra resolution of this technique. This figure also shows a spectrum (discrete points) for DNA damage to alfalfa seedlings. We have scaled the points for a maximum value of 1. The original careful quantitative work by Quaite and the Sutherlands at Brookhaven National Laboratory shows that outer layers shield the cells and reduce the sensitivity to UVB from that expected from data gathered from work on the susceptibility of unshielded plant cells.
CAUTION: Oriel® Solar Simulators are not designed for research on humans. Exposure to intense UV radiation can cause delayed severe burns to the eyes and skin.
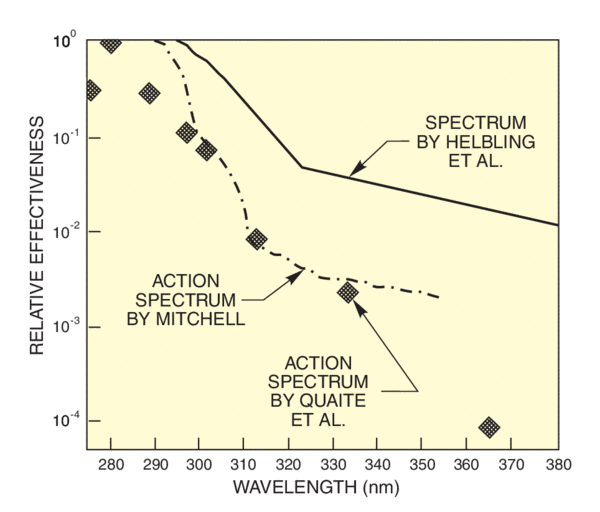
All the action spectra increase dramatically with decreasing wavelength. At first glance, any increase in UVB will lead to dramatic increase in DNA damage, erythema and inhibition of photosynthesis. Madronich7 used satellite measurements of ozone concentration from 1979 to 1989 to estimate the changes in UV reaching the earth's surface over this period. He used various action spectra to calculate the DNA and plant damage at various latitudes over this ten year period, estimating a 7.4% increase at 50 N and a 34% increase at 75 S. The difficulty in drawing conclusions, even from detailed measurements of ozone, lies in uncertainty of the key action spectrum. Madronich used Setlow's8 generalized DNA damage spectrum and Caldwell's9 plant damage spectrum. Recent detailed studies by Sutherland's group10 on human skin and plant seedlings casts some doubt on whether the potential increase in DNA damage due to higher UVB levels will be as high as predicted. They point out that much previous work underestimated the effects of UVA. Although the sensitivity of the effects to UVA is very low, UVA penetrates layers that shield the DNA much better than does UVB, and there is a lot more terrestrial UVA. Convolving Quaite and Sutherland's action spectrum for DNA damage in alfalfa seedlings with the relatively high level of UVA solar irradiation shows that the UVA contribution is significant. Since the levels of UVA will not change with ozone depletion, conclusions based on UVB increase, overestimate the impact of the loss of ozone. Additional data11 on UV damage to phytoplankton supports Sutherland's position.
References
- Diffey, B. Private Communication (1992-1993)
- Peak, J.G. and Peak M.J. Mutation Research, 246 (1991) 187-191
- Van der Leun Private Communication (1992-1993)
- Helbling et al. Marine Ecology Progress Series Vol 80:89-100, 1992
- Mitchell, B.G. and Karentz, D., Antarct. J. U.S. 26, 119-120, 1991
- Quaite, F.E., Sutherland, B.M. and Sutherland J.C. Nature Vol 358, p 576, August 1992
- Madronich, S. Geophysical Research Letters, Vol 19, No. 1, pp 37-40, 1992
- Setlow, R.B., Proc. Natl. Acad. Sci. 71 3363-3366, 1974
- Caldwell, M.M. et al. pp 87-111 Stratospheric ozone reduction, solar ultraviolet radiation and plant life, Worrest and Caldwell Editors, Springer Verlag, 1986
- Freeman et al., Proc Nat'l. Acad Sci. U.S.A., Vol 86 pp 5605-5609, July 1989
- Ryan, K.G.J. Photochem. Photobiol. B: Biol., 13 (1992) 235-240